If you're selling packaged food in both the US and the EU, 2026 is not a quiet year. The EU's new packaging regulation kicks in August. Anti-greenwashing rules follow in September. On the US side, the FDA is phasing out six synthetic dyes and states are writing their own rules faster than brands can track them. Two markets, four major changes, one year. Here's what your label and artwork teams need to know.
The fundamental difference you need to understand first
The FDA and EU don't just have different rules. They have different jobs.
The FDA's mandate is nutritional accuracy: is what's on the label true, and can consumers make informed choices based on it? The FDA is prescriptive and ingredient-focused. Rules get updated when science shifts or a public health issue reaches critical mass. The system is largely reactive.
The EU's mandate is broader: is the product, its packaging, and every claim on it defensible, environmentally and legally? The EU operates on a different premise entirely. The EU treats packaging as a policy tool. That's why compliance in Brussels feels more expansive than compliance in Washington.

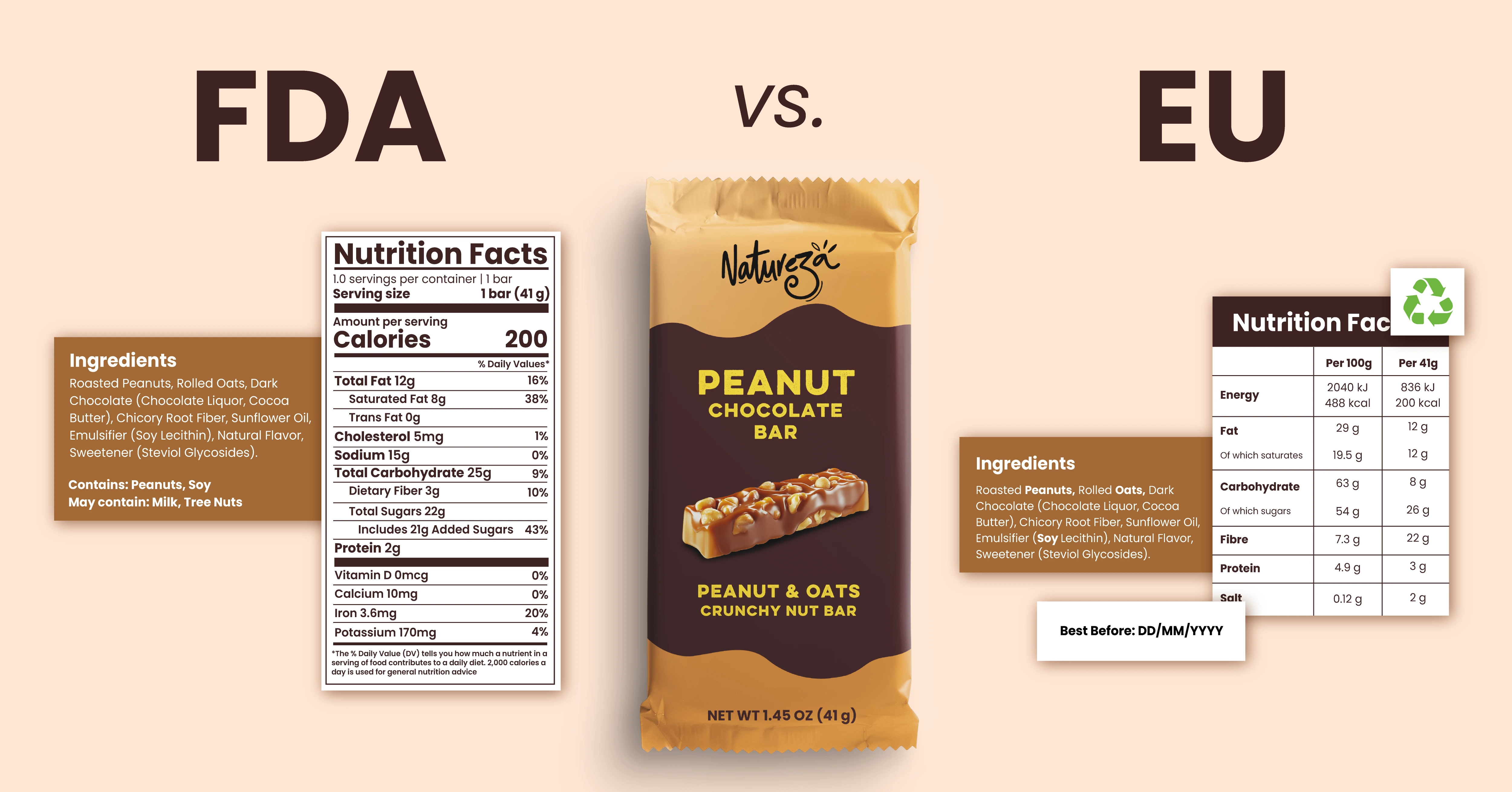
What's on the label: the baseline comparison
Both markets require the same core elements on pack, but how each is presented differs in ways that matter for cross-market label management.
What's actually changing in 2026 and what it means for your labels
In 2026, both markets are changing simultaneously, and the deadlines are closer than most teams realize.
In the US
Synthetic dye phase-out
In April 2025, the FDA announced a plan to eliminate six petroleum-based synthetic dyes Red 40, Yellow 5, Yellow 6, Blue 1, Blue 2, and Green 3 from the food supply, with the working timeline now set for end of 2027. The phase-out is voluntary at the federal level, not a binding rule — but the industry isn't waiting. The FDA is maintaining a public tracker of manufacturer's pledges, and several of the largest US food companies have already committed to completing reformulations by that deadline. If your products contain any of these dyes, your ingredient statement and color-related claims will need updating. Plan label artwork revisions now, not when reformulation is complete.
The FDA also issued enforcement discretion guidance allowing companies to use "no artificial colors" claims during the transition period, even before full reformulation is done, provided no FD&C-certified dyes remain in the product. That's a meaningful label claim opportunity, but it comes with its own substantiation requirements.
The state-level patchwork
Texas and West Virginia both enacted food additive labeling laws in 2025. Key details your team should know:
- West Virginia bans foods containing several synthetic dyes from retail shelves from January 2028
- Texas requires warning labels for certain ingredients, though the practical scope may be narrowed by federal preemption
- Over 140 food additive bills were introduced across 38 states in 2025
If your distribution spans multiple US states, you may already need market-specific label versions.
Front-of-Package Nutrition Info Box
Still a proposed rule, not finalized. The FDA is analyzing public comments and preparing options for leadership in 2026. If finalized, compliance timelines would be three to four years from the effective date. It's not an immediate action item, but brands redesigning packaging now should pre-engineer front panel real estate for it.
In EU
The PPWR is four months away
The Packaging and Packaging Waste Regulation (Regulation EU 2025/40) becomes legally binding across all 27 member states on 12 August 2026. For label and artwork teams, two things need to be on pack before that date: harmonized recyclability labels telling consumers which bin the packaging belongs in, and material composition pictograms. The Commission is still finalizing the exact pictogram formats, but the August deadline is fixed. After that date, non-compliant packaging cannot enter the EU market.
Later phases follow. QR code identifiers become mandatory from 2027, and full harmonized EU labeling from August 2028. But those timelines don't soften the first deadline. If your EU artwork hasn't been revised yet, August 2026 is the date to work back from.
The anti-greenwashing rule- September 2026
The EU's Directive on Empowering Consumers for the Green Transition (ECGT) takes effect on 27 September 2026. It bans generic environmental claims unless they are specifically substantiated with verifiable data. Practically, this means:
- Claims like "eco-friendly," "sustainable," "carbon neutral," and "nature-positive" are prohibited without proof
- Claims about future environmental performance require a detailed, publicly verifiable implementation plan
- Sustainability labels must be based on a recognized third-party certification scheme, not self-created
Any such language currently on your EU packaging needs a compliance audit before September.
PFAS in food contact packaging
Already prohibited from 12 August 2026 under the PPWR. If your packaging uses coatings, inks, or adhesives that contain PFAS, this isn't just a labeling change. It's a material change, and it may trigger a label revision if your compliance documentation references those materials.
Where brands most commonly get it wrong
Allergen labeling format
The US mandates declaration of eight major allergens. The EU mandates 14, adding molluscs, lupin, sulphites, and others. But the more common error isn't the count. It's the format. The EU requires allergens to be visually distinct within the ingredients list, typically bold or italics. The FDA allows a separate "Contains" statement below the ingredients. Brands copying a US label for EU markets frequently miss this distinction, and it's one of the most common compliance errors in cross-market launches.
Environmental claims on pack
In the US, sustainability language on packaging is largely voluntary and lightly regulated. In the EU from September 2026, it is legally scrutinized. A claim that packaging is "recyclable" must mean it is actually recyclable in the local collection infrastructure, not just technically recyclable in theory. "Made from sustainable materials" without certification is banned. What's acceptable on your US label may constitute a prohibited claim on your EU version of the same product.
What this means for your artwork workflow
Managing labels across the US and EU in 2026 isn't a one-time compliance project, it's an ongoing operational discipline. Each market now has its own regulatory timeline. A change triggered by the US synthetic dye phase-out shouldn't automatically touch your EU artwork. A PPWR update to EU recyclability icons shouldn't contaminate your US master file. That separation only holds if your artwork workflow manages each market's label as a discrete, versioned document with its own compliance status and approval history.
The brands that handle this well don't just have a compliance team, they have a system. When a regulatory change lands, they know exactly which SKUs are affected, which markets, and which label elements need to change. They can move from regulatory update to approved revised artwork without the process of creating downstream errors.
Your label audit checklist for both markets
Before your next packaging run, your team should be able to answer yes to all of these:
US
- All products using synthetic dyes have a reformulation plan and a corresponding label update timeline
- Front panel real estate has been assessed for a future FOP Nutrition Info Box
- State-specific requirements have been checked for every US market where the product is sold
- "No artificial colors" claims (if used) are supported by the February 2026 FDA enforcement discretion criteria
EU
- EU packaging artwork has been revised with recyclability and material composition labels ahead of 12 August 2026
- All sustainability claims on EU packaging have been audited against ECGT substantiation requirements ahead of 27 September 2026
- Allergen declaration is visually distinct within the ingredients list, not just a separate "Contains" statement
- PFAS-containing packaging materials have been reviewed and replaced where required
Staying compliant across two markets is not easy. ManageArtworks helps packaging and compliance teams do exactly that. See how it works.
Frequently Asked Questions
The core difference is philosophical. The FDA focuses on nutritional accuracy and ingredient transparency. The EU extends labeling responsibility to packaging sustainability, recyclability, and the substantiation of environmental claims. In practice, this means an EU-compliant label requires significantly more than its US equivalent, particularly from 2026 onward.
No. If you're selling in the EU, your product must comply with EU regulations regardless of where it's manufactured. FDA compliance alone is not sufficient for EU market entry. Brands selling in both markets typically maintain two separate label versions per SKU.
Each market's label should be treated as a discrete, versioned document with its own compliance status and approval history. When a regulatory change affects one market, it should trigger a review of that market's labels only, without affecting the other. This requires a structured artwork management workflow, not just a shared folder system.




.webp)
















